 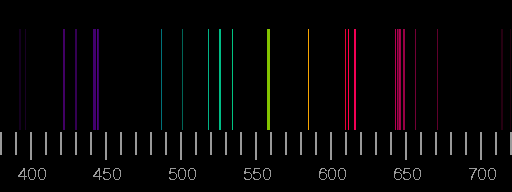
Symmetrical multiplets P - P, D - D, etc. The light which atoms give off is made up of specific wavelengths, called lines observed by a spectroscope, the lines are, collectively, atomic spectra. Relative Strengths for Lines of Multiplets in LS Coupling Normal multiplets S - P, P - D, D - F, etc. A discussion of their normalization as well as more extensive tables are given in Ref. . The total multiplet strengths S M are also listed in this table. The satellite lines y n and z n are usually weaker and deviate more from the LS values than the stronger diagonal lines when departures from LS coupling are encountered. Their strengths normally diminish along the diagonal. The strongest, or principal, lines are situated along the main diagonal of the table and are called x 1, x 2, etc. This table lists relative line strengths for frequently encountered symmetrical (P → P, D → D) and normal (S → P, P → D) multiplets in LS coupling. Relative Strengths for Lines of Multiplets in LS Coupling ISBN: 9780674834408.And n is the refractive index of standard air. Cambridge, MA: Harvard University Press, 1979. Cambridge, MA: Harvard University Press, 1963. In an emission spectra electrons are excited to an excited state by thermal or electrical means and then relax back to a lower state and emit a photon of light at a specific energy, which is seen as at a specific wavelength. ISBN: 9780521482516.īaade, W., and Cecilia Payne. There are two types of line spectra, emission and absorption. New York, NY: Cambridge University Press, 1996. When an electron transitions from one energy level to another, it emits light or photon with a specific wavelength. There are more energy states in an atom than there are electrons. Cecilia Payne-Gaposchkin: An Autobiography and Other Recollections. Nuclear Physics Atomic Spectra Atomic Spectra We know that in an atom, electrons have discrete and specific energies. This lecture gives more details about the atomic spectra of hydrogen along with matter/energy interactions involving atomic hydrogen. Explain how emission line spectra and absorption line spectra are formed. Lecture Slides (PDF - 1.3MB) Lecture Summary spectra of light emission using a diffraction grating. In this video, we'll use the Balmer-Rydberg equation to solve for photon energy for n3 to 2 transition. lines in the solar spectrum millions of such fixed absorption lines are now known. A line spectrum is a series of lines that represent the different energy levels of the an atom. Line spectra the Bohr model uses of emission and absorption spectra About Transcript The Balmer Rydberg equation explains the line spectrum of hydrogen. 6.3, “Atomic Spectra and Models of the Atom.” Describe the ground state of the gas phase atom.Īrchived Lecture Notes #1 (PDF), Sections 3, 5.Explain trends in ionization energy across the periodic table.Calculate the energy of an electron in the ground state of hydrogen.Explain why the radius of the electron orbital takes multiple values that are discrete, quantized, and non-linear.Describe how the atomic spectra of hydrogen is produced.Learning ObjectivesĪfter completing this session, you should be able to: Sadoway discusses the shell model and quantum numbers ( Session 5). Session 3: Atomic Models: Rutherford & Bohr.Drill down to an element and inspect the individual spectra lines. Sort by name, symbol, or count of spectra lines. Can search by element name, symbol, or atomic number. Matter/Energy Interactions: Atomic SpectraĪtomic spectra of hydrogen, matter/energy interactions involving atomic hydrogen, planetary model, Bohr’s postulates, quantum condition, ionization energy, electron orbital transitionsĪngstrom, Avogadro’s number, prism, refraction, wavelength, nanometer, Johann Balmer, wavenumber, Michael Faraday, cathode, anode, electron-volt, Bohr radius, ground state, ionization energy, energy level, conservation of energy, atomic spectra, Cecilia Payne, Ernest Rutherford, joule, coulomb, Max Planck, Planck’s constant, emission spectra, spectrograph, electrode, photon, volt, radiationĬhemical analysis, analyzing composition of stars, televisionīefore starting this session, you should be familiar with: Mac iPhone iPad Apple TV Reference app for Strong Line Atomic Emission Spectra in the visible range of 380-740nm. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
